Volume occupied by atoms hcp

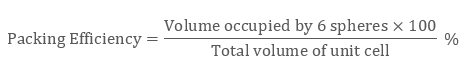
The total stimulation time represents only a fraction of the time scale of the friction process. The typical length of a simulation run is 10 4 time units which corresponds to a total simulation time of about 2 ns. The time scale of a solid friction process is estimated by the size of an average asperity and velocity, leading to a value of approximately 100 ns. Such high velocities are expected for example in case of thread breaking. Thus for the GEAM medal, the relative velocity is of the order of 30 m / s. In simulations presented here the relative velocity is about 0.01 c T ( the shear rate is ␥ ̇ = 0.001 ), where c T is the transverse sound speed. The dry solid friction process is characterized by large relative velocities at the interface. For most metals, the penetration hardness lies in the range 0.005–0.0025 ( reduced LJ units ) cf. Values for the isotropic pressure are estimated from the penetration hardness, defined as the ratio between load N and contact area A at onset of plastic flow. Thus it is important to use a sufficient large number of particles ͑ N Ͼ 5000 ͒ to obtain reliable information about dislocation formation, dynamics, and microstructure generation. Yield stress and the frequency of defects formed in the steady state shear regime depend via the energy needed to form a defect on the system size see Ref. We studied several system sizes to make sure that the presented results are not artificially driven by finite size effects. This results in a deviation between densely packed planes and planes normal to the flow gradient, potentially increasing the probability for the appearance of defects at finite size. Crystal planes tend to contain a multiple of unit crystal cells between the boundaries of the system. Boundary conditions potentially effect the newly formed crystal structure. For particular low rates, alternate simulation strategies, employing transient time correlation functions, evaluating differences between equilibrium and nonequilibrium trajectories, or beyond equilibrium molecular dynamics may be considered. One should note that NEMD simulations of sliding interfaces have a minimum shear rate below which computational costs become expensive.
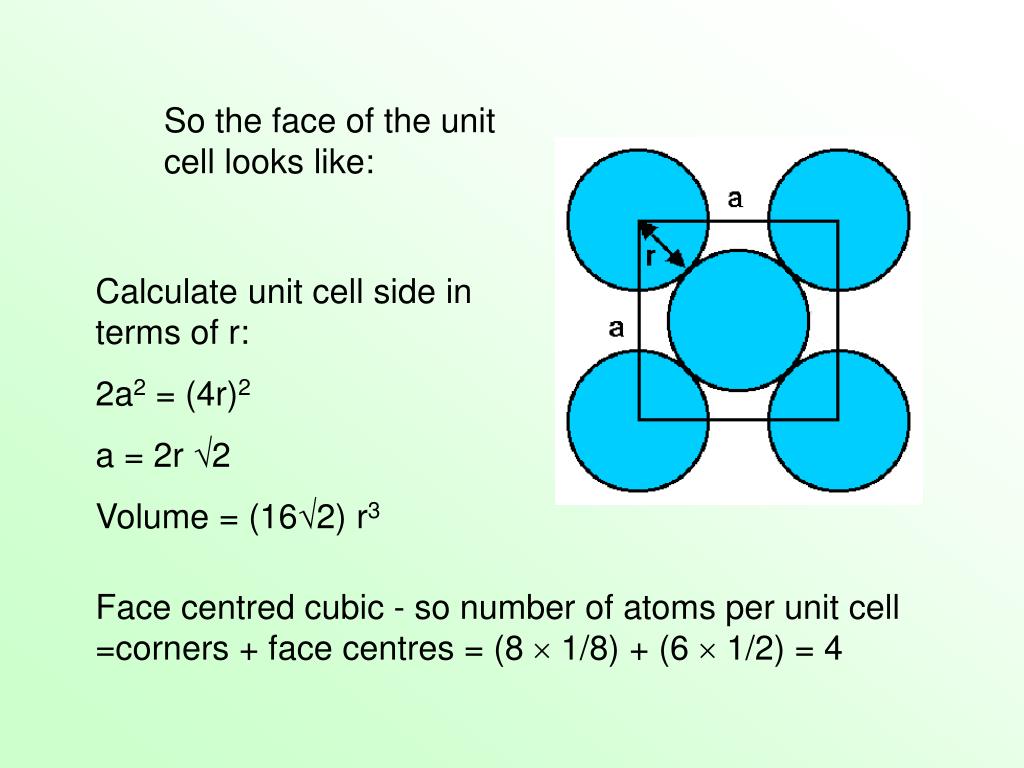
Alternatively, shear flow can also be generated by modifying the equations of motion with a SLLOD algorithm. Was this answer helpful?The correct order of the packing efficiency i¦see Refs. Hence, correct order is fcc (74%) > bcc (68%) > simple cubic (52%). Packing efficiencies of different cells are shown in the above table. What is the correct order of packing efficiencies of different cells? Packing fraction of different types of packing in unit cells is calculated below: Hexagonal close packing (hcp) and cubic close packing (ccp) have same packing efficiency. In this section we shall learn about packing efficiency. Percentage of spaces filled by the particles in the unit cell is known as the packing fraction of the unit cell. The packing efficiency of the simple cubic cell is 52. Since a simple cubic unit cell contains only 1 atom. What is the packing efficiency of a simple cubic cell? The unit cell that has the minimum or lowest Packing Efficiency is Simple Cubic with 52. Which unit cell has the minimum or lowest packing efficiency? Packing Fraction Formula = Volume Occupied by all constituent particles Total Volume of Unit CellUnit Cell Packing Efficiency – VEDANTUwww. Packing Efficiency Formula = No of Atoms x Volume Occupied by 1 atom Total Volume of Unit Cell X 100 We can say that Packing Fraction, when multiplied by 100, is seen as a percentage, it becomes the Packing Efficiency of that particular cell. The packing efficiency of simple cubic unit cell is 52. The packing efficiency of Bcc unit cell is 68%. The packing efficiency of Fcc unit cell is 74%. The packing efficiency is the percentage of total space occupied by the particles. What is the packing efficiency of simple cubic unit cell?

What is the packing efficiency of BCC and CCP? The volume occupied by 4 atoms is 4Ã-34‹Ïr3=316‹Ïr3 The packing efficiency =total volume of unit cellVolume occupied by atoms in a unit cell ‹Ã-100 The packing efficiency =88‹r3316‹Ïr3‹Ã-100=74. What is the packing efficiency of a CCP unit cell?Ī CCP unit cell has 4 atoms per unit cell.

